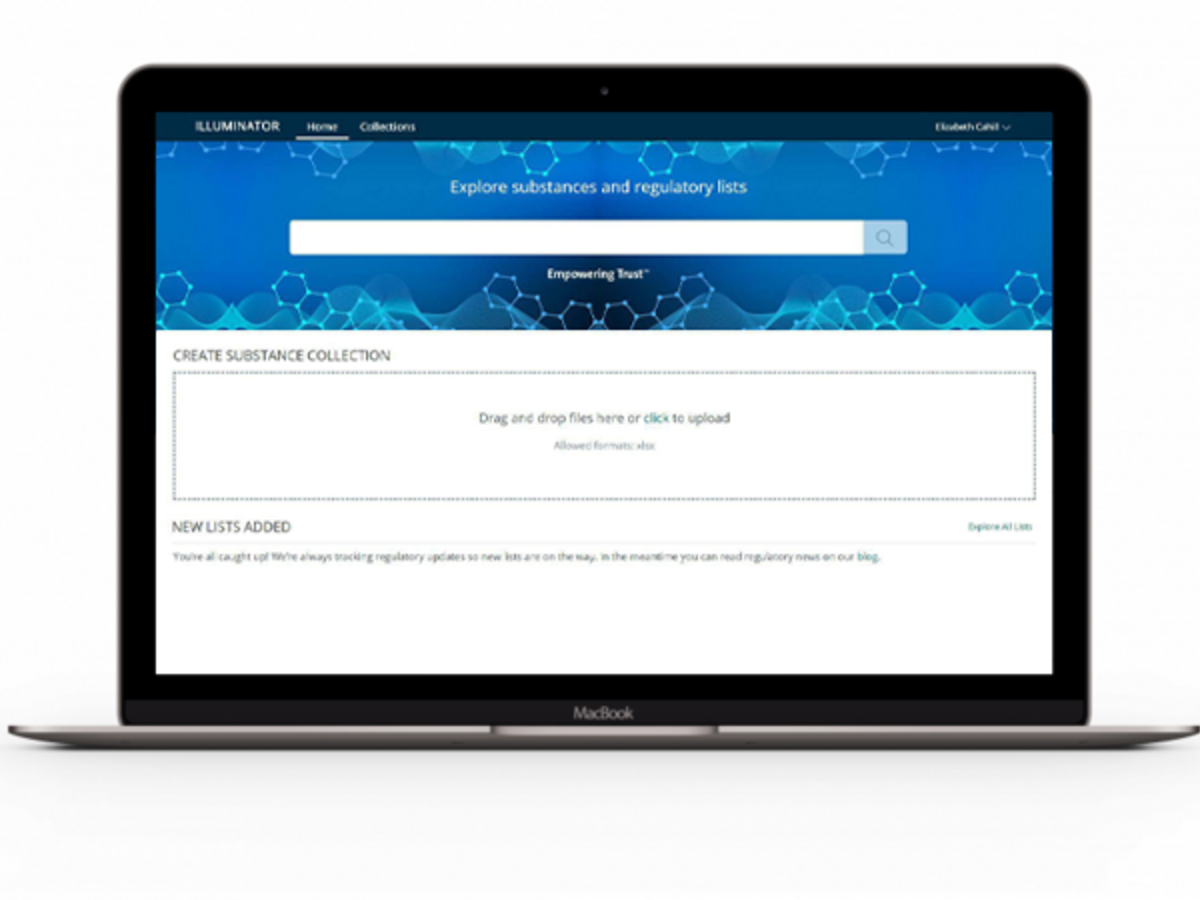
Quickly find information on chemical substances and regulations
The ChemADVISOR® Regulatory Database, formerly known as LOLI (Lists of Lists), helps you gain regulatory intelligence – containing over 8,000 regulatory lists from over 130 countries around the world to guide you in world-class chemical safety, product management, and environmental stewardship, including Safety Data Sheet and Label authoring and compliance with occupational, environmental, health and safety regulations.
It's never been more important for the data that drives your business to be trusted and up-to-date.
With the increasing speed and volume of regulatory change, and the complexity of supply chains, having access to current and trustworthy data is an integral part of your business.
As government and advisory agencies (as well as advocacy groups and brands) issue their policies in many languages and formats, which are then dispersed online and via various physical mediums, ChemADVISOR® is your single and structured source for the substance-specific data points you need for hazard assessment and product compliance around the world.
Providing the quality support you need
Hundreds of companies, including many household names and prominent government agencies, choose ChemADVISOR® to power their understanding of what’s in their products.
Our clients know that when it comes to compliance, cutting corners doesn’t pay.
Intelligent connections built-in
The information you need may be based on chemical families, a synonym or in a foreign language - but if you miss it, you’ll miss out. ChemADVISOR® is structured to link you to relevant data regardless of where you start your search.

Gain Global Market Access
Helping to ensure market access globally and for specific supply chain and retailers.
Substance and material identification
Identifying substances receiving attention from advocacy and advisory groups.
Compliance documentation
Creating required compliance documentation (safety data sheets, labels, and reporting).
Sustainability for your business
Includes sustainability, green chemistry and hazard assessment.
Powering your software with ChemADVISOR regulatory database
Datafeed
The database is delivered in SQL format via FTP download site.
The download comes with a full table mapping and description and is available in multiple formats.
API
The ChemADVISOR® API data access portal offers software developers RESTful web services to access ChemADVISOR® Regulatory Database.
The developer portal contains live documentation and copy/paste programming examples.
Our data supports a diverse set of industry sectors
Food and beverage

Personal care and cosmetics
Specialty and lab chemicals

Paints and coatings
Pesticides and biocides
Electrical and electronic equipment
Toys and children’s products
Textiles and clothing
Regulatory Roundup Newsletter
Stay up to date on chemical regulatory news
Subscribe to our monthly Regulatory Roundup Newsletter and stay up to date on current and upcoming regulations and all the latest chemical industry news.
Get connected with our team
Schedule a complimentary demo with a UL Solutions software expert today.